Source Of Energy :
- A source of energy is one which provides sufficient amount of energy in a convenient form over a long period of time .
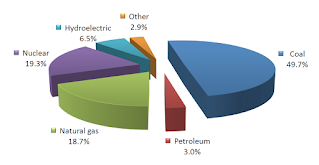
- Sources of energy are two types :
- (i) Non - renewable sources of energy (ii) Renewable sources of energy .
1 . Non - Renewable Sources of Energy:
- The sources of energy which are present in nature over a very , very long time and cannot be prepared when exhausted are called non - renewable sources of energy.
- These are present in the earth in a limited amount. they will not be prepared again when exhausted.
- For example , Fossil fuels ( Coal , Petroleum and Natural gas ) , and Nuclear fuels ( such as Uranium ) .
- The non-renewable sources of energy are also called conventional sources of energy.
2. Renewable Sources of Energy:
- The sources of energy which can be prepared continuously when exhausted are called renewable sources of energy.
- For example, Air, Water , Wood, Biogas , Alcohol , Hydrogen etc.
- The renewable sources of energy are also called non-conventional sources of energy.
- These sources of energy can be used again and again, endlessly.
- They are also called inexhaustible sources of energy.
Characteristics Of Source of Energy :
- it should do a large amount of work per unit mass
- it should cheap and easily available,
- it should easy to store and transport
- it should safe to handle and use
- it should not cause environmental pollution.
Fuels :
- The substances which produce heat energy on burning are called fuels. Examples : Wood, Coal, Cooking gas (LPG), Kerosene, Diesel, CNG etc.
- Heat energy is released when the fuels are burnt.
- All the fuels produce heat energy on burning.
- Some fuels produce more heat whereas others produce less heat.
- The usefulness of a fuel is measured in terms of its calorific value. Higher the calorific value, better the fuel will be.
Calorific Value :
- The amount of heat produced by one gram of fuel when burnt completely is known as its calorific value.
- e.g, when one gram of a charcoal is burned completely, it produces about 33 kilojoules of heat, so the calorific value of charcoal is 33 kJ/g per gram .
- calorific value of kerosene oil is 48 kJ/g. i.e . if 1 gram of kerosene oil is burnt completely, then it will produce 48 kilojoules of heat energy.
- Hydrogen is an extremely good fuel because of its high calorific value of 150 KJ/g. Since Hydrogen has the highest calorific value, therefore, a fuel containing higher percentage of hydrogen will have a higher calorific value. For example, LPG , Methane etc.
Ignition Temperature :
- The minimum temperature required at which a fuel catch fire to produce heat is known as its ignition temperature.
- No fuel can burn unless it is heated to its ignition temperature .
Characteristics Of An Ideal Fuel Or Good Fuel:
- It should have a high calorific value.
- It should burn without giving out any smoke or harmful gases.
- It should have a proper ignition temperature . i.e. ignition temperature should neither be too low nor too high. if the ignition temperature of the fuel is very low, then the fuel will catch fire too easily and hence it will be very unsafe to use it. On the other hand, if the ignition temperature is too high, then it will be very difficult to light the fuel.
- It should be cheap and easily available.
- It should be easy to handle, safe to transport, and convenient to store.
- It should not leave much ash behind after burning.
- It should burn smoothly.

No comments:
Post a Comment